
Safe & Simple
Pain Relieffor diabetic nerve pain
FDA-cleared for the symptomatic relief of chronic, intractable pain from Diabetic Peripheral Neuropathy
A wearable prescription device for chronic nerve pain
NeuMona is an FDA-cleared, wearable prescription medical device indicated to provide symptomatic relief of chronic, intractable pain from diabetic peripheral neuropathy. It is designed to be comfortably worn on one ear for a seven-day treatment period and then removed. NeuMona is prescribed and applied by a qualified healthcare professional.
NeuMona does not cure diabetes or neuropathy. It is intended to help reduce pain symptoms and support improved daily function.
When diabetic nerve pain starts to control your life
Patients often describe the challenge of diabetic peripheral neuropathy as:
“It feels like my feet are on fire at night. As soon as I lie down, the pain starts and I can’t sleep.”
“Simple activities have become a challenge. Every step reminds me something is wrong.”
“I want to play with my grandkids, but I’m afraid of losing my balance or making the pain worse.”
“The constant discomfort has become exhausting.”
Why standard treatments can fall short
Incomplete or inconsistent pain relief
Side effects such as dizziness, sedation, or cognitive fog
Medication interactions or tolerance over time
Long-term reliance on drugs without durable improvement

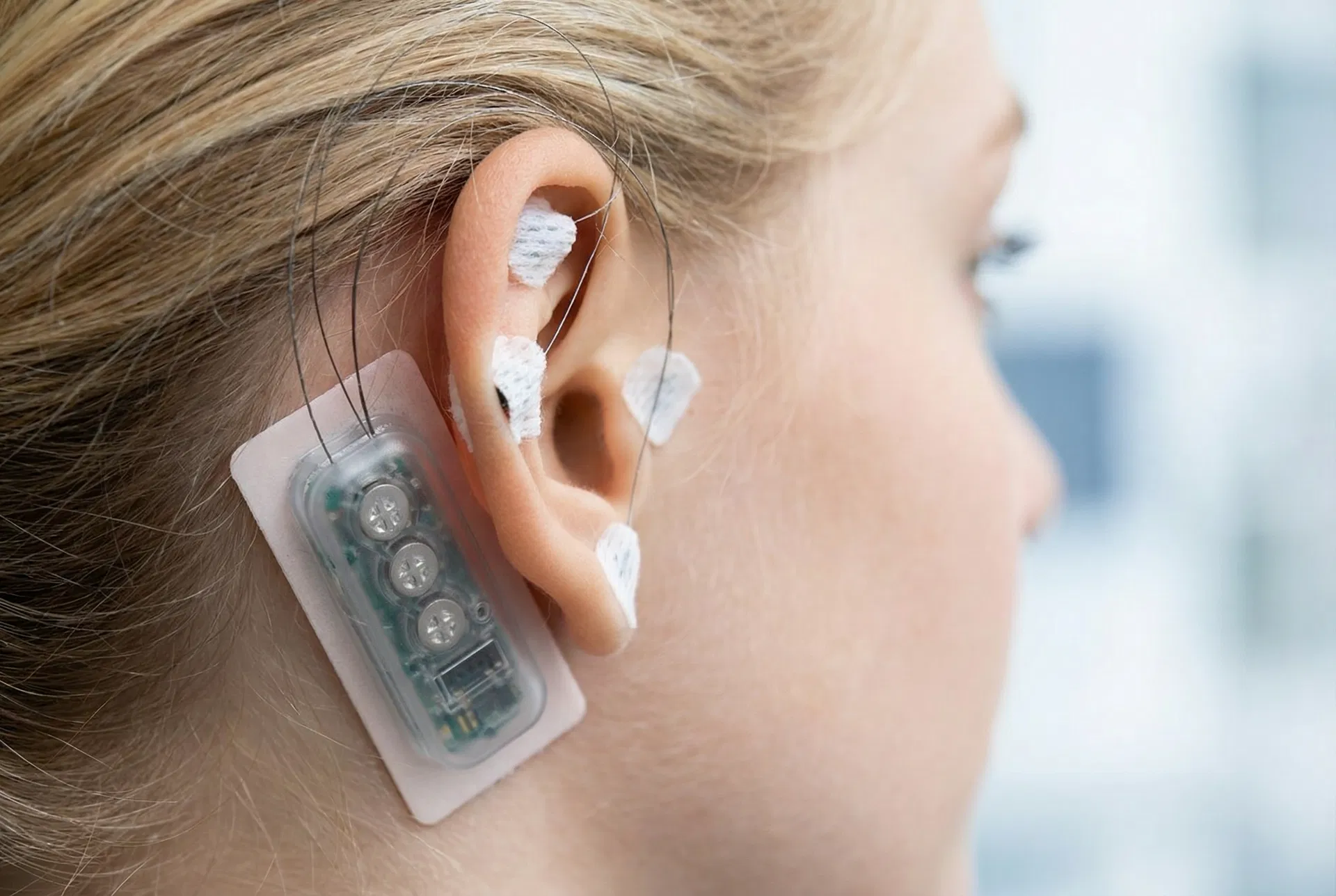
NeuMona device
A different approach to pain
Delivers gentle electrical signals through specific nerve branches in the outer ear, including the vagus nerve, that are connected to pathways involved in pain perception and sensory processing
Worn casually with minimal impact on daily life and activities
Medication-free and non-addictive — no drugs enter your body
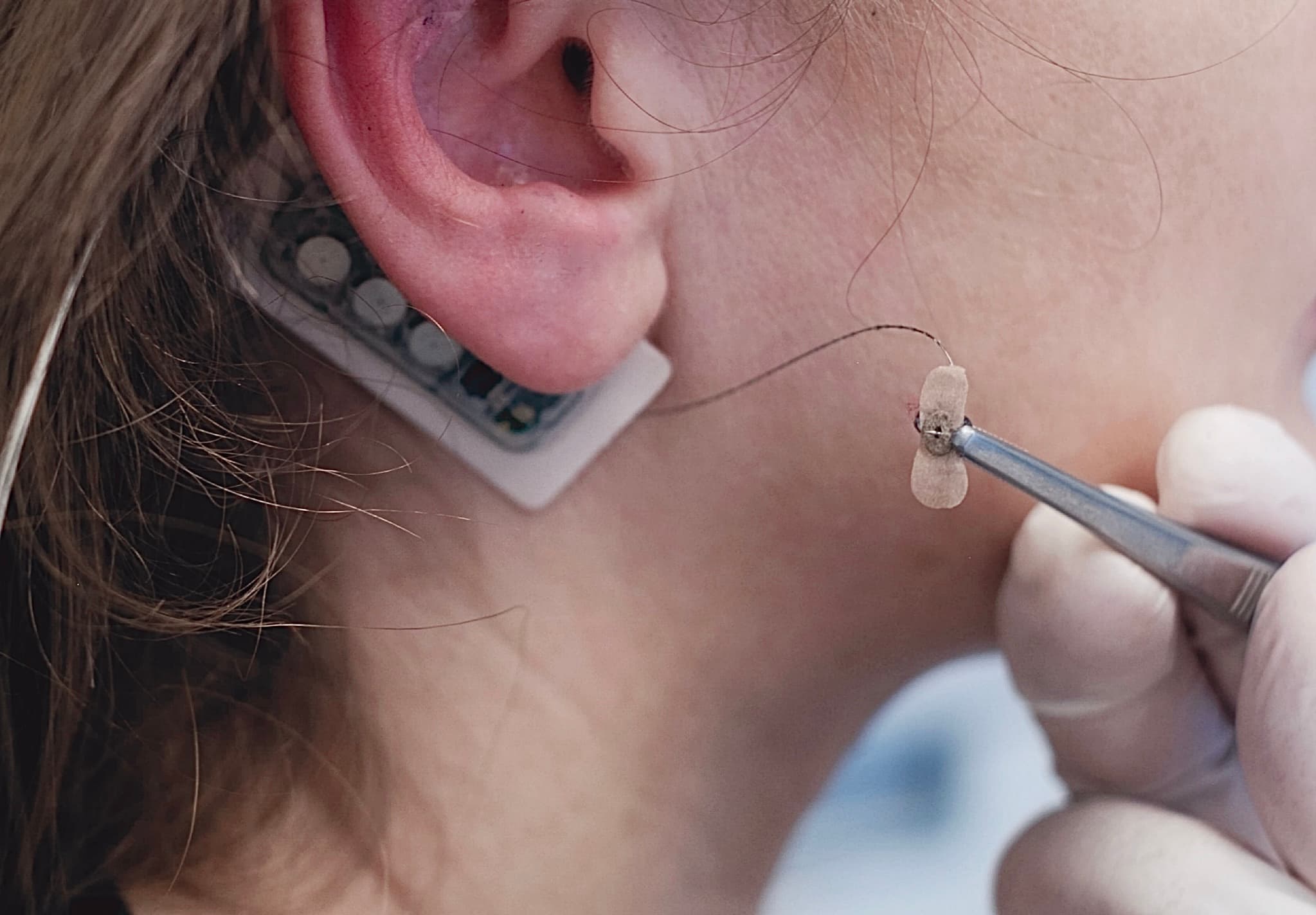
Non-surgical and minimally invasive — four tiny microneedles placed on the outer ear
Simple, brief in-office application without anesthesia
The Device
Small, discreet, comfortable

What patients commonly report
Improvement in pain intensity
Improved sleep quality
Improved physical function and ability to perform daily activities
Reduced anxiety related to chronic pain
Improved nerve sensitivity in the feet
Reduced need for additional pain medications
Individual results may vary.
Madhuchander S, Gurunath S. Comparing Percutaneous Electrical Neurostimulation with Placebo in the Management of Diabetic Peripheral Neuropathic Pain. 2019. ClinicalTrials.gov Identifier: NCT03540446.
What to expect
Consultation
A licensed healthcare provider evaluates the patient and determines if NeuMona is appropriate.
In-Office Placement
No anesthesia required. Four tiny microneedles are placed on the outer ear.
7-Day Wear
Wear NeuMona at home while you work, sleep, and live normally.
Removal & Follow-Up
Device is removed in-office. Multiple treatment cycles may be used as directed by your clinician.

Helping patients get back to the moments that matter.
Questions about NeuMona
NeuMona is a prescription medical device. This webpage is for informational purposes only and does not provide medical advice. Patients should consult a licensed healthcare provider to determine whether NeuMona is appropriate for them. Distributed by MAI Medical USA Inc.
Ready to explore NeuMona?
Find a trained provider near you, or learn how to offer NeuMona in your practice.